Is air a homogeneous or heterogeneous combination? This elementary query delves into the intricate composition of the air we breathe, revealing a captivating interaction of gases and their distribution. Understanding the reply is not nearly chemistry; it unlocks insights into the very ambiance that surrounds us and shapes our world.
The ambiance, a posh mix of gases, performs an important function in sustaining life on Earth. Nitrogen, oxygen, argon, and hint quantities of different gases mix to create the air we breathe. Figuring out if this combination is homogeneous or heterogeneous hinges on whether or not its elements are uniformly distributed at a microscopic stage.
Defining Homogeneous and Heterogeneous Mixtures
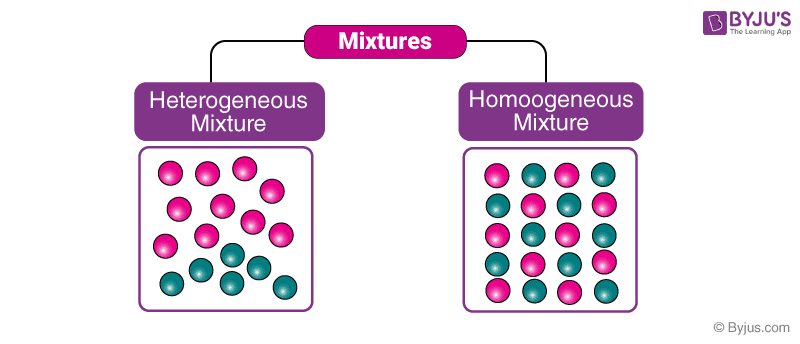
Understanding the distinction between homogeneous and heterogeneous mixtures is key to comprehending the properties and conduct of matter. These classifications are essential in numerous scientific and sensible purposes, from meals processing to materials science. Figuring out the traits that outline every kind permits for a deeper understanding of the substances we encounter each day.Homogeneous and heterogeneous mixtures differ basically of their composition and look.
A radical understanding of those distinctions is crucial for correct evaluation and prediction. This part delves into the important thing traits that distinguish these two kinds of mixtures, offering examples and a comparative desk to help comprehension.
Defining Homogeneous Mixtures, Is air a homogeneous or heterogeneous
Homogeneous mixtures exhibit a uniform composition all through. This implies the elements are evenly distributed at a molecular stage, making the combination seem visually uniform. The person elements can’t be distinguished with the bare eye or fundamental commentary.
Defining Heterogeneous Mixtures
Heterogeneous mixtures, in distinction, show a non-uniform composition. Their elements should not evenly distributed, and the totally different elements of the combination are simply distinguishable. This lack of uniformity is obvious within the combination’s look.
Examples of Homogeneous and Heterogeneous Mixtures
A wide range of substances exemplify every kind. Saltwater is a homogeneous combination, the place salt dissolves utterly in water, leading to a uniform resolution. Air, a combination of gases, additionally represents a homogeneous combination. In distinction, a combination of sand and water is a heterogeneous combination; the sand particles stay suspended within the water and are clearly seen.
Equally, a salad is a heterogeneous combination of varied substances.
Traits of Homogeneous and Heterogeneous Mixtures
| Characteristic | Homogeneous | Heterogeneous |
|---|---|---|
| Composition | Uniform | Non-uniform |
| Look | Uniform | Non-uniform |
| Particle Dimension | Uniform (or dispersed to the molecular stage) | Non-uniform |
The desk above summarizes the important thing variations between homogeneous and heterogeneous mixtures. The uniformity of composition, look, and particle dimension is vital in distinguishing them.
Particle Distribution in Mixtures
The distribution of particles considerably impacts the traits of the combination. In homogeneous mixtures, particles are dispersed on the molecular stage, making a constant composition all through. In heterogeneous mixtures, particles should not evenly distributed, usually remaining separate entities or phases inside the combination. This distinction in particle distribution is an important think about figuring out whether or not a combination is homogeneous or heterogeneous.
Air as a Combination
Air, the life-sustaining envelope surrounding our planet, is a posh combination of gases. Understanding its composition is essential for comprehending its function in climate patterns, local weather change, and even human well being. This intricate mix of components dictates the very air we breathe. Comprehending the make-up of air offers insights into its properties and behaviors.The ambiance, a dynamic system, consists primarily of nitrogen and oxygen, with smaller portions of different gases.
This various mixture creates the air we depend on. The relative proportions of those gases considerably affect the ambiance’s traits and interactions with the Earth’s floor. These proportions aren’t static; they fluctuate based mostly on numerous elements.
Composition of Air
The composition of air isn’t uniform; it varies with altitude and different elements. Understanding the important thing elements and their relative abundances is significant. Air is primarily composed of nitrogen and oxygen.
- Nitrogen (N 2): Roughly 78% of the ambiance is nitrogen. Its inert nature makes it an important part for sustaining the soundness of the ambiance.
- Oxygen (O 2): Essential for respiration, oxygen includes about 21% of the ambiance. This very important gasoline is crucial for all times as we all know it.
- Argon (Ar): A noble gasoline, argon accounts for roughly 0.93% of the ambiance. Its presence contributes to the general composition and properties of the air.
- Hint Gases: Different gases, together with carbon dioxide (CO 2), neon (Ne), helium (He), methane (CH 4), and krypton (Kr), exist in a lot smaller portions however play vital roles in numerous atmospheric processes.
Distribution of Gases in Air
The distribution of gases within the ambiance is not uniform. The density of gases decreases with altitude. Essentially the most dense elements, like nitrogen and oxygen, are concentrated nearer to the Earth’s floor. 
Diagram exhibiting the reducing density of gases as altitude will increase. The very best focus of gases is discovered close to the Earth’s floor, and the density regularly thins out as you progress larger into the ambiance.
Formation of Air
The formation of air is a posh course of tied to the Earth’s formation and evolution. The early ambiance was drastically totally different from the one we all know right this moment. Volcanic outgassing, asteroid impacts, and organic exercise have all contributed to the present composition of air.
Air Stress and Elements
Air strain is a drive exerted by the load of the air above. The strain at sea stage is considerably larger than at larger altitudes as a result of higher mass of air urgent down. The totally different densities of the atmospheric gases contribute to the general strain gradient.
Strategies of Separating Air Elements
Numerous strategies might be employed to separate the elements of air. These strategies usually depend on the totally different boiling factors and different bodily properties of the gases. The commonest methodology is fractional distillation.
- Fractional Distillation: This course of includes cooling the air to extraordinarily low temperatures. The liquefied air is then allowed to heat regularly. Completely different elements boil off at various temperatures, permitting them to be collected individually. This course of takes benefit of the variations in boiling factors of gases in air.
Movement Chart of Air Separation
A movement chart detailing the method of separating air elements utilizing fractional distillation.
- Compression: Air is compressed to extend its density.
- Cooling: Compressed air is cooled to liquefy it.
- Fractional Distillation: The liquefied air is slowly heated. Completely different gases vaporize at totally different temperatures, permitting for his or her assortment.
- Assortment: Collected gases are saved based mostly on their boiling factors.
Properties of Air Elements
The properties of air’s elements affect its conduct and affect on the surroundings. Understanding these properties is vital for predicting and managing atmospheric phenomena.
- Nitrogen: Inert and unreactive, nitrogen offers a steady ambiance.
- Oxygen: Important for respiration, oxygen helps combustion and is significant for all times.
- Argon: A noble gasoline, argon is unreactive and utilized in numerous purposes.
- Hint Gases: These gases play important roles in atmospheric processes, just like the greenhouse impact.
Air’s Homogeneity/Heterogeneity: Is Air A Homogeneous Or Heterogeneous
Air, a seemingly easy substance, is definitely a posh combination of gases. Understanding its composition and the way it behaves below totally different situations is essential for numerous purposes, from climate forecasting to aerospace engineering. This examination delves into the nuances of air’s homogeneity, exploring the elements that affect its obvious consistency and the the explanation why it is categorized as a homogeneous combination regardless of its various elements.A elementary facet of understanding air is its composition.
Air is predominantly nitrogen and oxygen, with hint quantities of different gases like argon, carbon dioxide, and water vapor. The distribution of those elements performs a pivotal function in figuring out air’s properties. This intricate combine offers rise to the query of whether or not air is really a homogeneous or heterogeneous combination.
Evaluating Air’s Composition to Different Mixtures
A key to understanding air’s homogeneity is evaluating it to each homogeneous and heterogeneous mixtures. Homogeneous mixtures, like saltwater, have a uniform composition all through. In distinction, heterogeneous mixtures, like a salad, exhibit distinct elements that aren’t uniformly distributed. Air, regardless of containing numerous gases, shows a uniform distribution of its elements at a given location and time.
This uniformity is a defining attribute that differentiates it from a heterogeneous combination.
Traits Indicating Air’s Classification
Air’s constant composition at a given location is a defining attribute. The continual mixing of gases within the ambiance ensures a uniform distribution of elements. This fixed mixing course of, pushed by wind and different atmospheric phenomena, maintains a constant composition over giant spatial scales.
Why Air is Thought-about Homogeneous
Regardless of the varied gases inside air, the minute focus of particular person elements permits for a constant total composition. That is essential for the uniformity that makes air a homogeneous combination. The vastness of the ambiance and the fixed mixing mechanisms make sure the constant proportion of gases. Air’s uniform composition, at any given level, is a testomony to the blending course of.
Components Affecting Air’s Homogeneity
A number of elements can have an effect on the homogeneity of air, notably on smaller scales. Native climate patterns, comparable to localized temperature variations or air pollution occasions, can result in non permanent variations in composition. Nevertheless, these localized fluctuations don’t alter air’s total homogeneity on a bigger scale.
The Mixing Course of and Air’s Composition
The fixed mixing of gases within the ambiance is the important thing to air’s homogeneous nature. Wind, turbulence, and different atmospheric phenomena drive this mixing, making certain a uniform distribution of elements. This dynamic course of is crucial for sustaining the steady composition that defines air as a homogeneous combination.
Arguments for and In opposition to Classifying Air as Homogeneous
| Argument | Rationalization |
|---|---|
| For | The continual mixing of gases ensures a uniform distribution of elements. |
| For | Air’s composition stays comparatively fixed over giant spatial scales. |
| In opposition to | Localized air pollution occasions or temperature inversions can create non permanent variations in composition. |
| In opposition to | The presence of various gases in air could appear to contradict homogeneity. |
Components Influencing Obvious Homogeneity
The obvious homogeneity of air is considerably influenced by the size of commentary. On a worldwide scale, air’s composition stays comparatively fixed, whereas on a neighborhood scale, short-term variations can happen as a result of localized situations. These variations, whereas noticeable, don’t negate the general homogeneity of air.
Air’s Habits Below Numerous Circumstances
| Situation | Description |
|---|---|
| Excessive Altitude | Air density decreases, and the focus of some gases, like oxygen, might differ barely as a result of decreased strain. |
| Low Altitude | Air density will increase, and the composition is comparatively uniform as a result of continued mixing. |
Epilogue

In conclusion, whereas air seems uniform and constant, its molecular make-up, and the interactions between its elements, are key to understanding its homogeneity. The constant proportions of gases in air, regardless of the complexity of its formation and interactions, make it a primary instance of a homogeneous combination. Understanding these elements is significant for appreciating the soundness and significance of our ambiance.
Key Questions Answered
What are the principle elements of air?
Air is primarily composed of nitrogen (roughly 78%), oxygen (roughly 21%), and argon (roughly 1%). Hint quantities of different gases, comparable to carbon dioxide, neon, and helium, are additionally current.
How does air strain relate to the composition of air?
Air strain is a results of the load of the air molecules urgent down on a floor. At sea stage, air strain is larger as a result of higher weight of the overlying air column. The composition of air, nevertheless, stays comparatively fixed all through totally different altitudes, regardless of the modifications in strain.
Can the elements of air be separated?
Sure, the elements of air might be separated utilizing numerous strategies, comparable to fractional distillation. This course of exploits the variations in boiling factors of the gases to isolate them.
How does air behave at totally different altitudes?
Air strain and density lower with growing altitude. The composition of air, nevertheless, stays comparatively fixed, regardless of the modifications in density and strain. At larger altitudes, there could be a slight variation within the focus of sure gases as a result of atmospheric dynamics, however total homogeneity is maintained.




